Sugar-free and low caloric sweetener
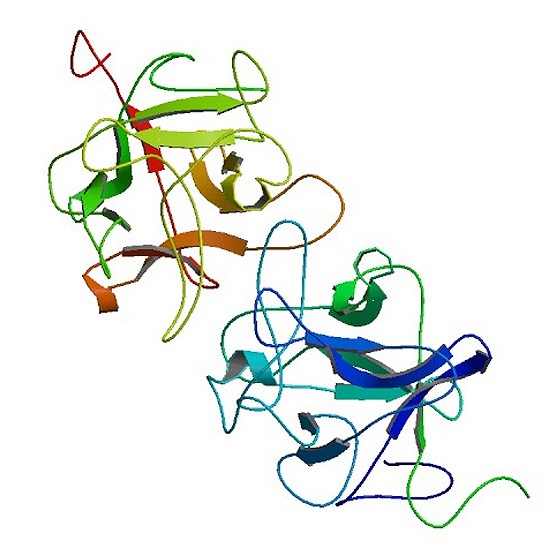
Figure 1: Representation of miraculin (https://www.rcsb.org/structure/3IIR).
Sugar that is mainly obtained from sugarcane and sugar beet is used to sweeten foods and drinks. The search for sugar-free and low caloric sweetener to replace sugar is especially relevant these days. The aim of our project is to develop a low caloric sweetener to replace sugar and thus contribute to reduce the increase of obesity. For that, we want to use the so called miraculin berry, which was discovered in the 18th century and is growing on the Synsepalum dulcificum plant. Miraculin (Figure 1) is a homodimeric, glycosylated protein that is flat in taste at neutral pH but that exhibits a taste-modifying activity to convert sour stimuli to sweetness under acidic conditions (pH 6.5 to 4.8). This modification of the taste occurs because the protein activates hT1R2-hT1R3 receptor (Figure 2) (Koizumi et al., 2011).
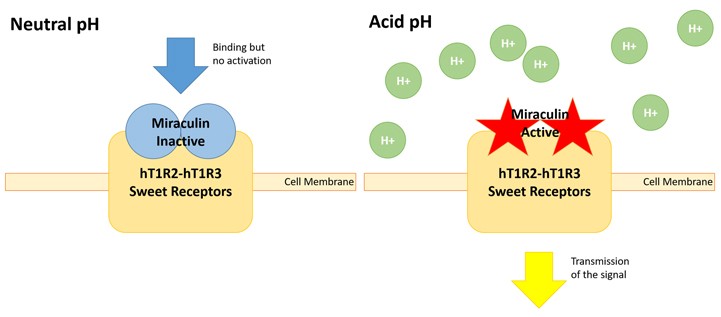
Figure 2: Scheme of the effect of the miraculin in the sweet receptors hT1R2 and hT1R3.
The appropriate bioreactor
However, Synsepalum dulcificum has a low productivity and is difficult to cultivate outside of its natural environment (Ezura & Hiwasa-Tanase, 2018). For this reason extracting the protein from the plant would end up in an expensive product. In order to increase the production, we decided to produce the miraculin in a different organism: We chose Spirulina.
Spirulina (Figure 3) is the common name used to define the biomass of the cyanobacteria Arthrospira platensis and Arthrospira maxima. Cultivated worldwide, Arthrospira sp. is used as a dietary supplement or even as food itself. Spirulina has a very fast growing rate and can be easily cultivated in open ponds or in closed bioreactors.
Figure 3: Image of Spirulina in a ZEISS Axio Imager 2.
Dried, it contains about 60% of proteins, including all the essential amino acids, and mainly saturated and polyunsaturated fatty acids. It also has a high mineral content, especially iron, magnesium, potassium, phosphor and it has a lot of vitamins and antioxidants, especially beta-carotene. That being said, by expressing miraculin in Arthrospira platensis it will not only result in a higher production of miraculin but also in a healthy and sweet food supplement.
Despite the great advantages of spirulina, there are limited successful strategies for genetic engineering of spirulina and some complications have been reported. The main reason for this failure is the presence of multiple strong cytoplasmic nucleases that can cleave foreign DNAs. Different studies have found a great number of restriction enzymes in spirulina strains (Kawamura et al., 1986; Tragut et al., 1995).
Using Agrobacterium for transformation
Fortunately, in a study from 2018 a new system that allows the engineering of Arthrospira platensis with good results has been found. Dehghani et al. (2018) used Agrobacterium tumefaciens, a gram-negative bacterium that causes gall tumours in plants. The bacterium is used for the stable transformation of a very large group of different cells, in plants, fungi, microalgae, and even human cells in the field of bioengineering.
Our approach to engineer spirulina is by using pCAMBIA1304 (Figure 4), a plasmid that can be introduced in Arthrospira platensis via the T-DNA of Agrobacterium. We have applied some modifications to the plasmid in order to increase the specificity to our strain of Arthrospira by introducing homologous regions. These are used to achieve a better recombination of miraculin and incorporate different promoters, terminators and marker genes. We also have included a strep-tag sequence in the miraculin gene in order to facilitate the purification of the protein after the expression so we can easily check if the spirulina is producing the protein.
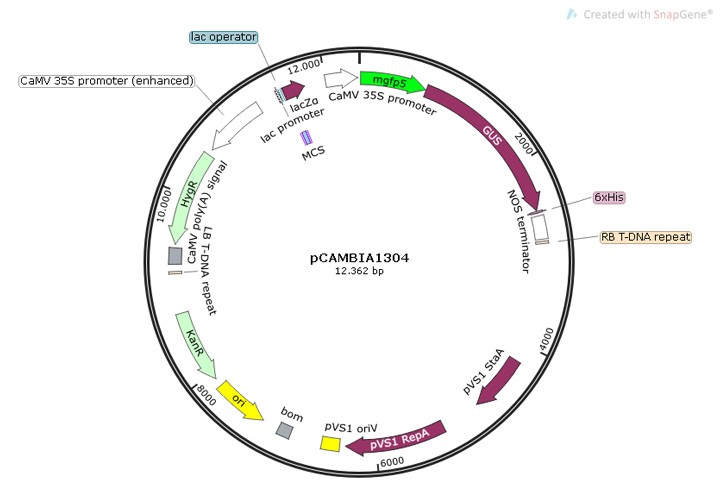
Figure 4: Map of the pCAMBIA1304 from SnapGene.
Finally, we have prepared a feeding assay with Drosophila melanogaster to test if the flies can feel the sweetness of the cyanobacteria. In this assay, normal spirulina and engineered spirulina that are stained with different dyes are administered and after 16 hours it is tested which spirulina is eaten more by the flies.
With this project we expect to create a not only a way to increase the production of miraculin but also participate in the establishment of new standards for the engineering of spirulina.
Our Team
This project is part of this year’s iGEM competition from the team of TU Dresden.

From left to right – Upper row: Behbood Moeini, Victoria Sarangova, Sophie-Luise Heidig, Nikitha Vavilthota, Juan Sebastian Eguiguren and Anastasia Kurzyukova – Lower row: Mara Müller, Pedro Guillem, Paula Santos Otte, Arnau Pérez Roig
By Arnau Perez Roig and Dr Andreas Ebertz
Did you like this article? Then subscribe to our Newsletter and we will keep you informed about our next blog posts. Subscribe to the Eurofins Genomics Newsletter.






One Comment