DNA Day fact #1: The powerhouse of DNA condensation
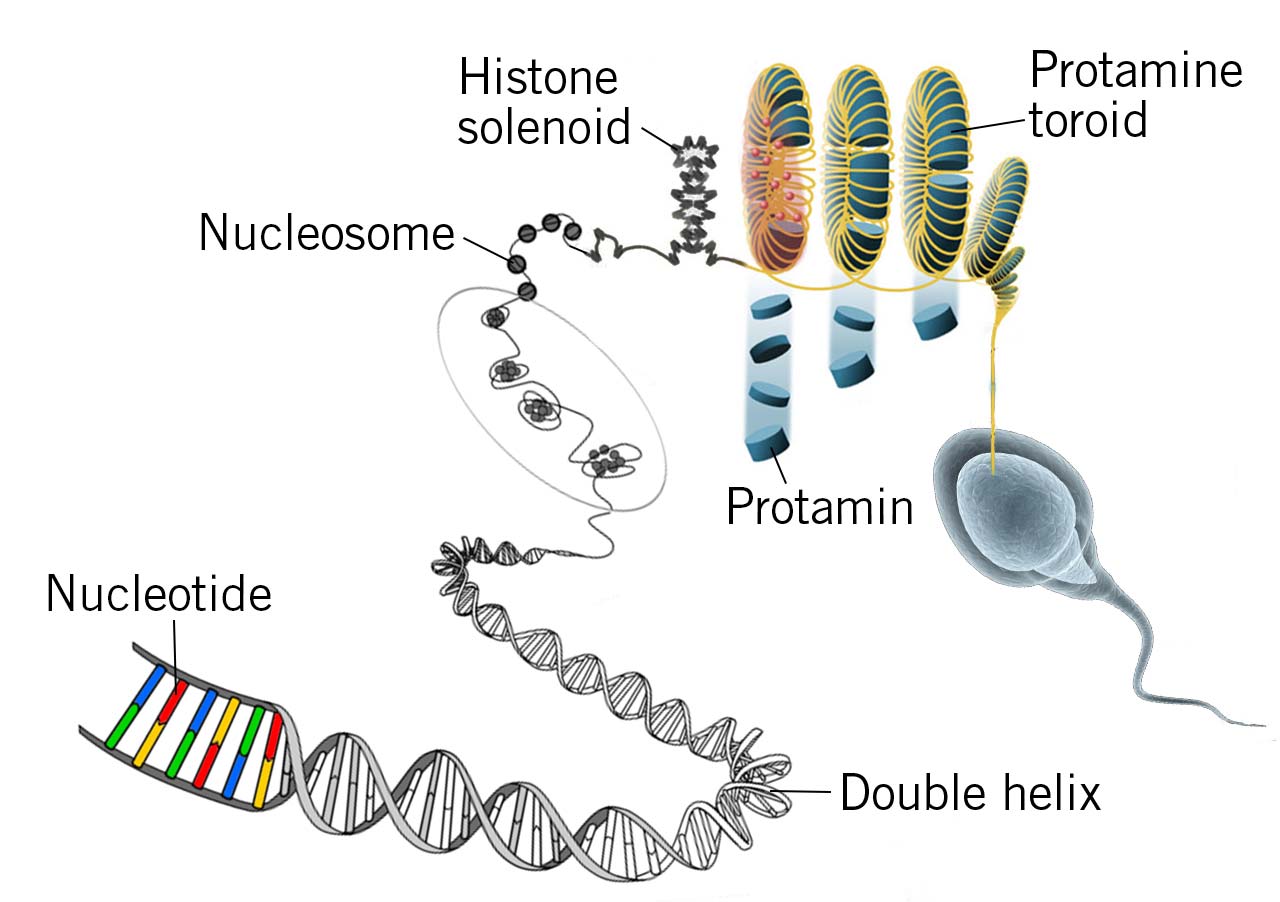
One of the first pieces of information that everyone learns during their first lesson on DNA is that a segment of DNA is wrapped around eight histone proteins to form the basic structure unit known as a nucleosome.
Intriguingly, this is not true for all cell lines. In fact, mammals have a special type of cell, in which nucleosomes are significantly different. Male germ cells, that are haploid, undergo numerous transformations during the late phase of the spermatid development; flagellum acquisition is just the most visible one. The paternal genome is entirely reorganised into a new condensed state via the substitution of the histone proteins with protamines.
Protamines are basic proteins rich in lysine and cysteine residues that allow extreme condensation. Most mammals have one protamine gene, but in humans and mice, there are two genes, clustered on the same chromosome (Bao and Bedford, 2016). The DNA–protamine complex is super-coiled into toroids, structures six to twenty times more condensed than the “standard” solenoids – true powerhouses in the field of miniaturisation!
DNA Day fact #2: NETs – the Neutrophil Kamikaze
Neutrophils are the most abundant white cells in our blood and are known as the first line of defence against acute bacterial infection. Even though their most known bactericidal mechanism is de-granulation (the release of antimicrobial molecules from vesicles inside the cell called granules), they can also form Neutrophil Extracellular Traps (NETs) in a last-ditch effort. Dying neutrophils decondense their chromatin and extrude their DNA complexed with histones and neutrophil elastase (serine proteinase secreted during inflammation) to ensnare bacteria. DNA can damage bacteria due to the phosphodiester bond of its backbone, which is a strong chelator of divalent cations (Sørensen and Borregaard, 2016). NETs are also equipped with anti-microbial peptides and proteases with a potent bactericidal effect, and neutrophils are not afraid to use it to kill their “sworn enemies” (Boeltz et al., 2019).
When grounded and out of munition, they think: “I am the munition” and sacrifice themselves.
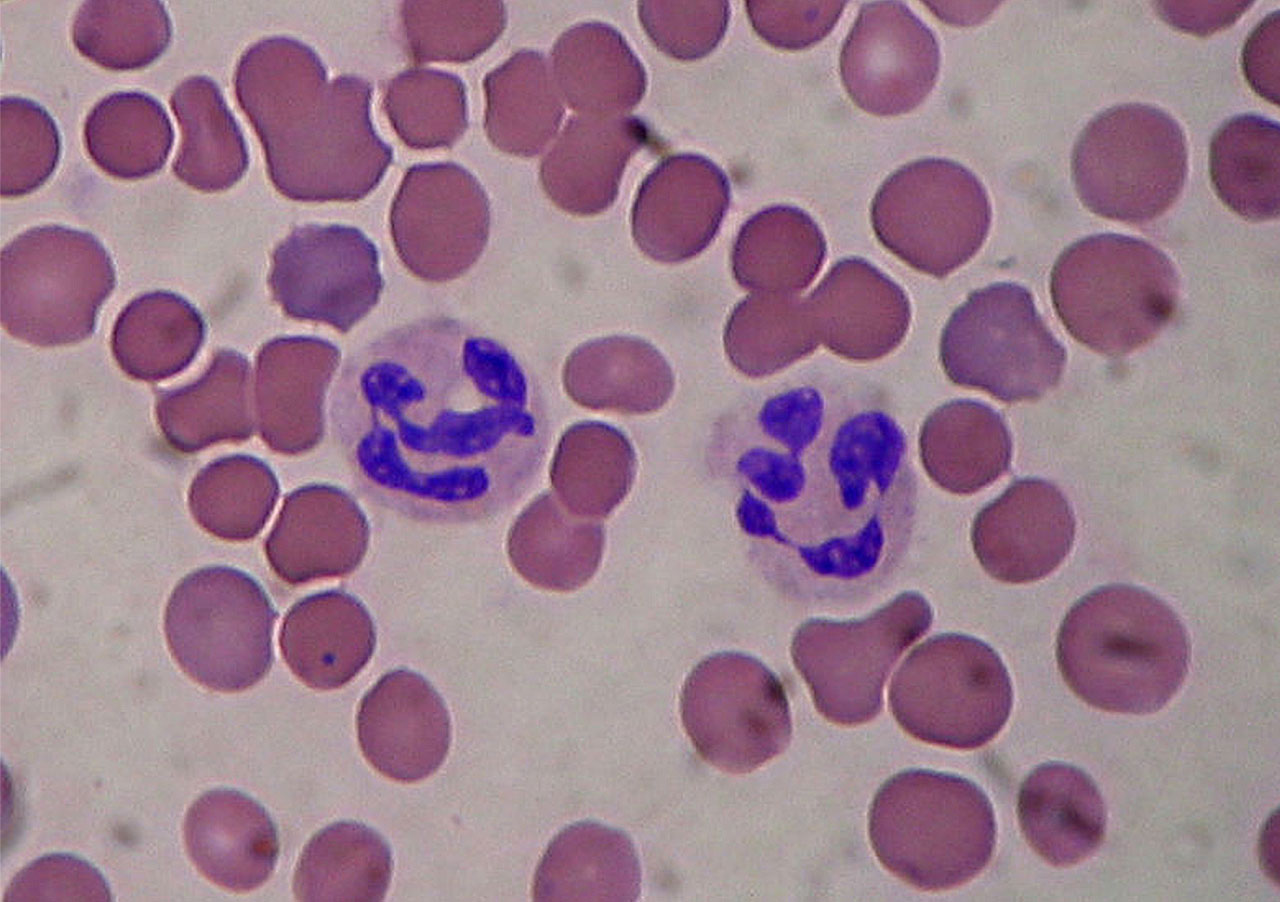
Image: NETs and the schematic process of their function (created in BioRender.com)
DNA Day fact #3: Furry friend or marvellous manifestation of epigenomics?
In their early embryonal stage, female placental mammals undergo a process called “lyonisation” (named after English geneticist Mary Lyon), which causes cells from the late blastocyst stage to randomly inactivate one of their two X chromosomes. This results in the formation of two cell populations made up of the descendants of those embryonal cells; two lines that make every mammalian adult female a mosaic.
The colouration of the tortoise shell cat is evidence of this phenomenon: the fur-colour gene happens to be located on the X chromosome. Depending on the embryonal derivation, two subsets of cells express either the black or the orange allele.

Image: Tortoise shell cat
DNA Day fact #4: Encoding a movie in the genomes of living bacteria
What if DNA was used as data storage?
Actually, this is precisely the main function of DNA, except the nature of the stored information can be very unexpected. For example, Seth Shipman and his team, presumably movie lovers, succeeded in encoding a digital short film into the genomes of living bacteria. Using the CRISPR-Cas system, they first converted the pixels into a nucleotide code. Subsequently, the oligonucleotides were integrated into the genome by bacteria over-expressing the Cas1-Cas2 integrase complex. By high-throughput sequencing, the original movie was reconstructed (Shipman et al., 2017).
This experiment sounds very futuristic and makes the imagination run wild, but probably nobody would want to be in the shoes of the bacteria that will have to store the entire saga of Game of Thrones.
Fun fact: The used video of a galloping horse is one of the first short films ever called The Horse in Motion by Eadweard Muybridge from 1878.

Figure: Encoding a short-film in bacteria (Shipman et al., 2017).
DNA Day fact #5: The big mitochondrial exception – like father like son
Two dogmas revolve around mitochondria: Firstly, the mitochondrion is the powerhouse of the cell and secondly, the mitochondrial DNA (mtDNA) is exclusively inherited by one’s mother. However, the second dogma has recently been called into question by Dr. Shiyu Luo and colleagues.
The story began with a boy who had a suspected mitochondrial disease. Researchers performed high resolution mtDNA sequencing and found no disease-related mutations, but an intriguing fact: the boy had a really high level of heteroplasmia, which refers to the coexistence of different mtDNA molecules in the same cell. Extending the analysis further, this unusually high heteroplasmia was found both in his mother and two healthy sisters. To uncover the mystery of this odd mtDNA pattern, the researchers investigated the boy’s maternal grandparents. They found that this peculiar mtDNA pattern was a product of mtDNA from both grandparents. This case, along with another similar report provide evidence of biparental mtDNA inheritance in humans (McWilliams and Suomalainen, 2019).

DNA Day fact #6: DNA Day… and Malaria Day!
Another very special commemoration is also tied to the 25th of April: World Malaria dDay!
The link between World Malaria Day and DNA Day is that, reportedly, a wide range of genetic variants seems to confer significant protection against death from malaria. Speculation on this began many years ago, when JBS Haldane proposed thalassemia (blood disorders with reduced haemoglobin production) as a survival advantage against malaria. Later, the frequency of haemoglobin S (related to sickle cell anaemia) in people from malaria endemic areas prompted a strong positive correlation in a similar way. Other polymorphisms of red blood cells seem to occur in malaria-affected populations. This is particularly relevant because Plasmodium parasites (etiologic agents of malaria) are known to first infect the human liver and, later, red blood cells. The asexual reproduction cycle of Plasmodium leads to an exponential multiplication of parasites in the blood, with serious consequences for the human host. Characterising how these genes confer protection could eventually help in the development of novel therapeutic approaches to fight malaria (Kariuki and Williams, 2020).
Did you know any of these facts? Which is your favourite? Let us know in the comments below! Stay tuned to the DNA Universe blog for more interesting content and have a great DNA Day.
By Valeria Di Leonardo, Giovanni Leucci and Dr Andreas Ebertz
Did you like this article on the DNA Day? Then subscribe to our Newsletter and we will keep you informed about our next blog posts. Subscribe to the Eurofins Genomics Newsletter.





